Description
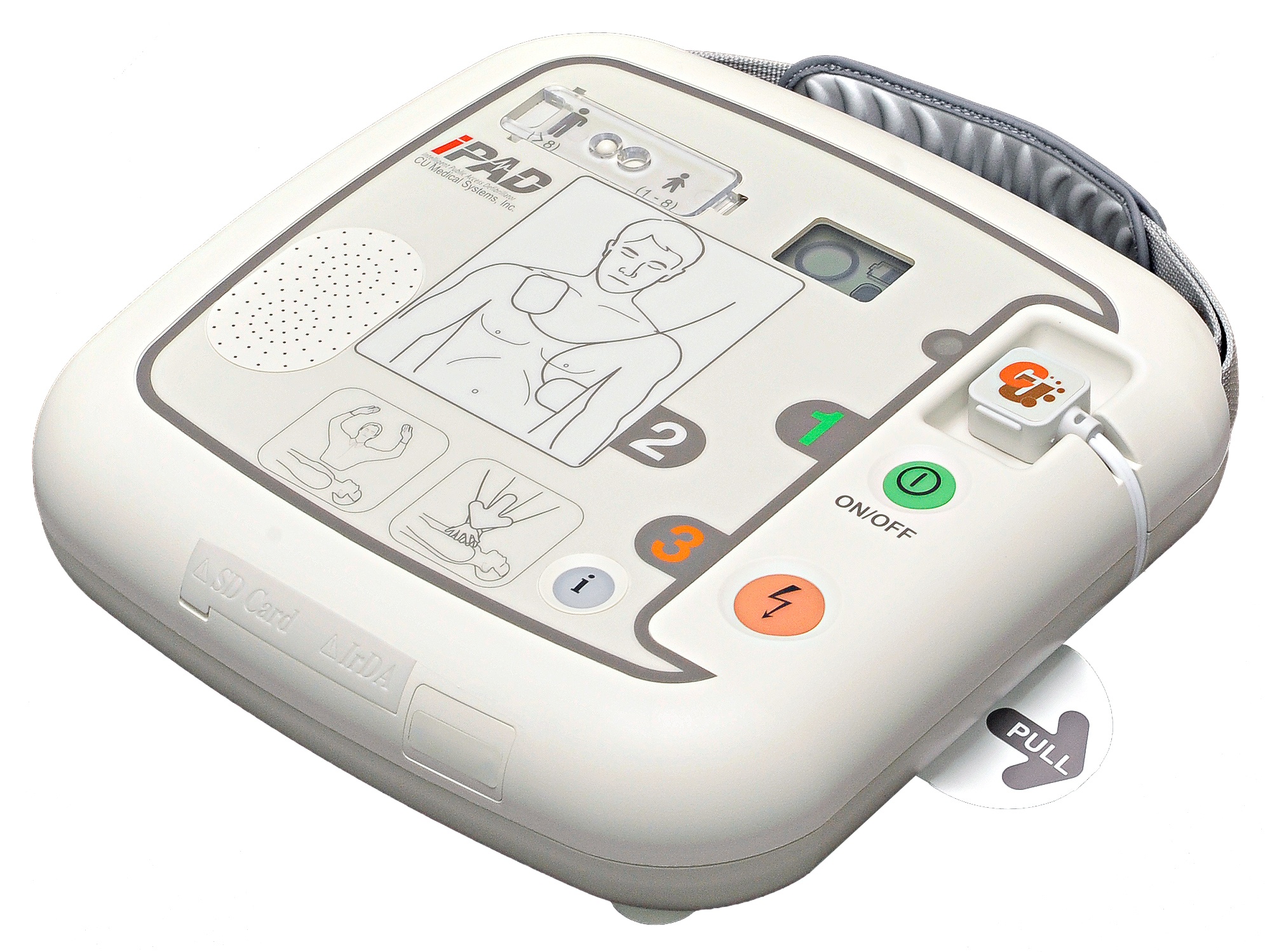
This device is a semi-automated external defibrillator that can be administered on sudden cardiac arrest (SCA) patients. CU-SP2 is an easy-to-use Semi-Automated External Defibrillator (AED) that is small, light, and portable, and uses a battery. The AED automatically or manually reads the sudden cardiac arrest (SCA) patient’s electrocardiogram (ECG) and determines if a cardiac arrest that requires defibrillation has occurred, so that licensed emergency medical technicians, medical professionals and the general public can easily operate it. SCA can occur anytime to anyone at any place and may threaten the patient’s life if the appropriate CPR and/or electric shock with a defibrillator are not applied within a few minutes.
Functions and Features
Ambient noise detection [Auto volume adjusting]
Easy-to-use & manage Electrode (Displaying Life expectancy of Electrode)
Graphic LCD Status Indicator (Device, Pads & Battery)
Selectable Voice guide for both Lay Rescuer and Healthcare Provider
ECG monitoring or multi-printing via Bluetooth communication
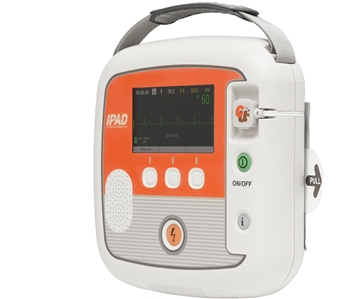
Product Exterior
Dimension: 260 x 256 x 70 mm (Width x Length x Height)
LCD: 5inch wide, 480×272 16bit
Weight 2.4? (Including the battery pack and pads).
Defibrillator
Operation Type: Dual Mode (semi-automated, manual) External Defibrillator
Output Type: E-Cube Biphasic (Truncated exponential type) Output Energy
AED Mode: 150J±4J, 200J±6J at 50? load for adults, 50J±2J at 50? load for children
Manual Mode (Optional): 2J±1J, 3J±1J, 5J±1J, 7J±1J, 10J±1J, 20J±2J, 30J±2J, 50J±2J, 70J±2J, 100J±4J, 150J±4J, 200J±6J at 50? load.
Self-Diagnostic Test
Auto Power Self-Test / Real-time Self-Test
Daily / Weekly / Monthly Self-Test
Manual Battery Pack Self-Test (performed when the user inserts the battery pack)
ECG Analysis System
Analyzes whether the rhythms of the patient’s impedance and ECG require a defibrillation
Measured Impedance Range 25? ~ 175?
Battery
Battery Type: 12V DC, 111.1V DC, 1.9Ah Li-ion, Rechargeable
Capacity: For fully charged new batteries, at least 60 shocks or 3 hours of operation at 25°C (77°F)
Standby Life (After Inserting the Battery): If stored and managed in accordance with instructions in the document: At least 2 years from the date of installation into the i-PAD CU-SP2
.
Temperature Ranges for Storage and Use
Operating Environment: 0°C ~ 43°C (32°F ~ 109°F)
Storage Environment: Temperature: -20°C ~ 60°C (-4°F ~ 140°F).
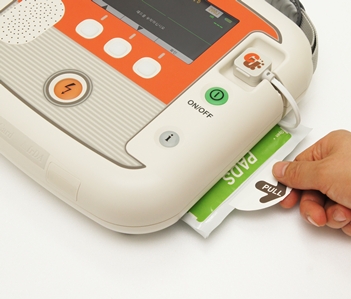
Electrode
Adult Electrode
Area: 110 cm2
Cable Length: Total 120cm (Internal: 95cm, External: 25cm)
Pediatric Electrode
Area: 50 cm2
Cable Length: Total 120cm (Internal: 80cm, External: 40cm)
Data Storage and Transfer
Data Storage: Saves 3 events on the internal memory (up to 17 hours per event)
SD Card: Stores the ECG and event data from the device’s internal memory through the PC software (CU-Expert)
Bluetooth: Uses Bluetooth to communicate with the Printer or the CUEM1 (ECG transmission device)
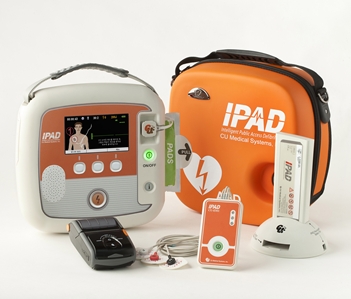
Standard Package
Device(1), Adult Pads(1), Rechargeable Battery (1), Battery Charge Dock(1), CU-EM1(1), Printer(1), Printer Paper(10), SD Card(1), CU-Expert (1), Carrying Case(1), User’s Manual(1)
Optional Accessories
Pediatric pads(1), Disposable battery(1), IrDA Adapter(1)